
PUBLICATION HIGHLIGHTS
Anand Vaidya, Amir Hamrahian, Irina Bancos, Maria Fleseriu, Hans K. Ghayee, on behalf of the AACE Pituitary, Gonad, Adrenal, and Neuroendocrine Disease State Network. The evaluation of incidentally discovered adrenal masses. Endocr Pract. 2019;25(2):178-192.
The prevalence of incidentally discovered adrenal adenomas has grown substantially in recent years due to an increase in cross-sectional imaging. Clinicians are faced with the challenge of evaluating these adrenal masses to prevent future health hazards and risks. Incorrect or insufficient evaluation of adrenal adenomas could put patients at an increased risk for preventable adverse cardiometabolic outcomes. Therefore, the American Association of Clinical Endocrinologists developed a Disease State Clinical Review to simplify the general approach to incidentally discovered adrenal masses with a case-based overview that builds on previous guidelines and consensus statements. Although not formal guidelines, these recommendations provide clinicians with a practical approach to the evaluation of incidentally discovered adrenal masses.
THERE ARE 2 FUNDAMENTAL QUESTIONS THAT CLINICIANS ARE FACED WITH WHEN DISCOVERING AN ADRENAL MASS:
Clinicians should consider biochemical screening for adrenal hormone excess, even if radiographic characteristics suggest that the adrenal mass is benign. In most scenarios, patients do not display the typical signs of overt syndromes of hormone excess, such as Cushing syndrome. However, clinicians may still uncover hormone excess upon biochemical evaluation.
THE FOLLOWING ADDITIONAL QUESTIONS MAY ARISE IN CERTAIN PATIENT SITUATIONS:
- Would a biopsy be useful in the diagnosis, management, and prognosis of the adrenal mass?
- Is medical treatment or surgery warranted?
- Could long-term surveillance of the adrenal mass, including biochemical testing and imaging, be useful? If so, for how long and at what frequency?
Managing incidentally discovered adrenal masses
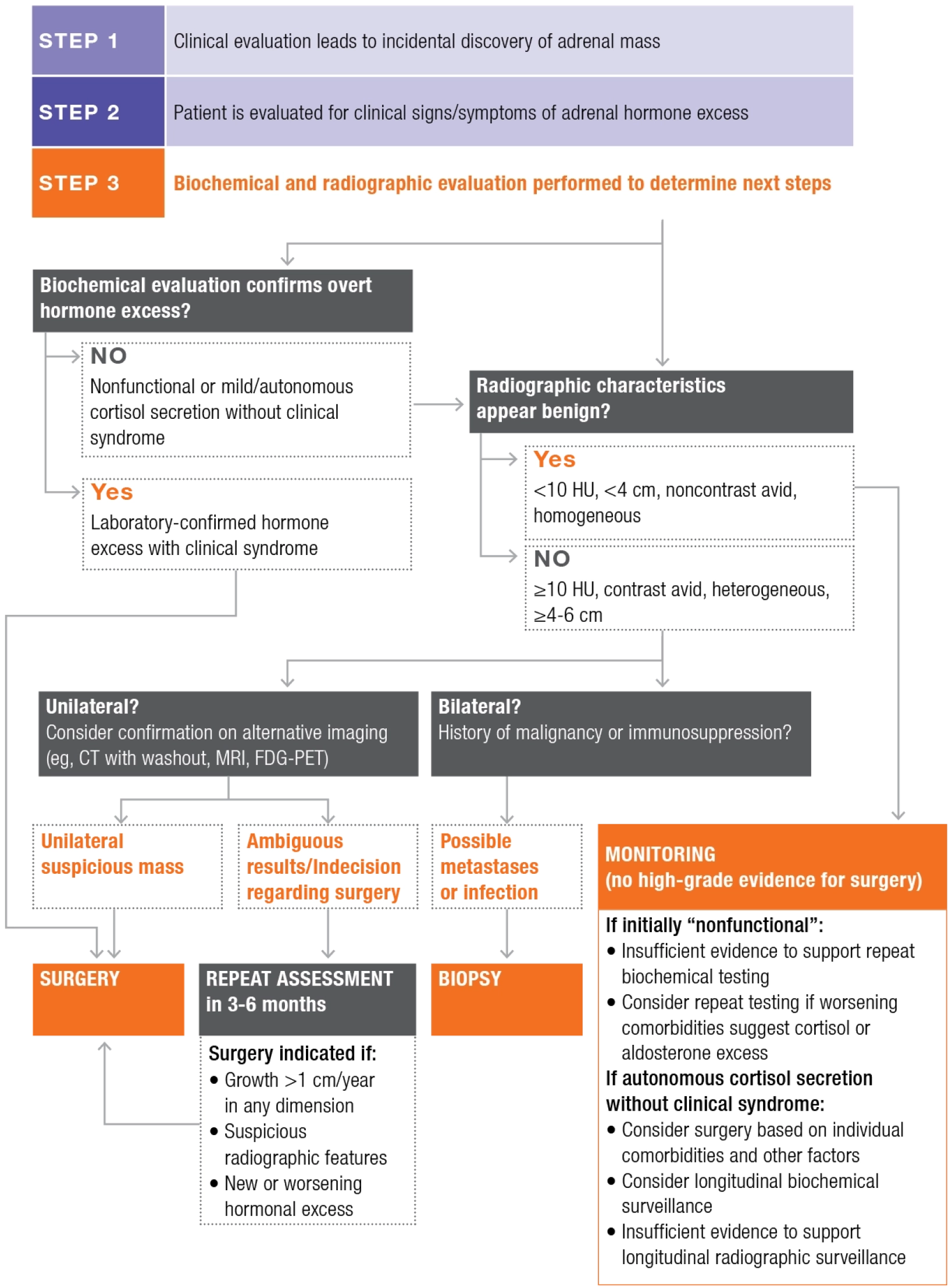
CT=computed tomography; FDG-PET=fluorodeoxyglucose-positron emission tomography; HU=Hounsfield units; MRI=magnetic resonance imaging.
Based on information from Vaidya et al., Endocr Pract. 2019.
Guidelines for biochemical testing of adrenal hormone excess in patients with incidentally discovered adrenal mass
| Suspected diagnosis | Autonomous cortisol secretion | Primary aldosteronism | Pheochomocytoma | Adrenal androgen excess |
|---|---|---|---|---|
| Who to test | All patients | Patients with high blood pressure and/or low potassium | Patients whose imaging shows lipid-poor, contrast- avid, heterogeneous adrenal lesions | Patients with abnormal hair growth, symptoms suggestive of virilization |
| Evaluation | 1-mg overnight dexamethasone suppression test | Serum ARR | Plasma (or urinary) fractionated metanephines | DHEAS total testosterone |
| Abnormal result | Nonfunctional: ≤1.8 µ/dL Possibly functional: 1.9-5.0 µg/dL Autonomous hypercortisolism: >5.0 µg/dL | Suppressed ARR >20-30 | Greater than 2 to 4 times higher than the upper limit of the reference range | Higher than the upper limit of the reference range |
Who to test
All patients
Patients with high blood pressure and/or low potassium
Patients whose imaging shows lipid-poor, contrast-avid, heterogeneous adrenal lesions
Patients with abnormal hair growth, symptoms suggestive of virilization
Evaluation
1-mg overnight dexamethasone suppression test
Serum ARR
Plasma (or urinary) fractionated metanephrines
DHEAS total testosterone
Abnormal result
Nonfunctional:
≤1.8 μg/dL
Possibly functional:
1.9-5.0 μg/dL
Autonomous hypercortisolism:
>5.0 μg/dL
Suppressed ARR >20-30
Greater than 2 to 4 times higher than the upper limit of the reference range
Higher than the upper limit of the reference range
ARR=aldosterone-to-renin ratio; DHEAS=dehydroepiandrosterone sulfate.
Based on information from Vaidya et al., Endocr Pract. 2019.
- The late-night salivary cortisol test and the 1-mg overnight dexamethasone suppression test (DST) are the most sensitive tests for detecting subtle forms of autonomous cortisol secretion and nonphysiologic cortisol secretion
- DST may be repeated after a few months to evaluate for consistently elevated cortisol levels
- The 24-hour urinary-free cortisol test is frequently normal in mild autonomous cortisol excess